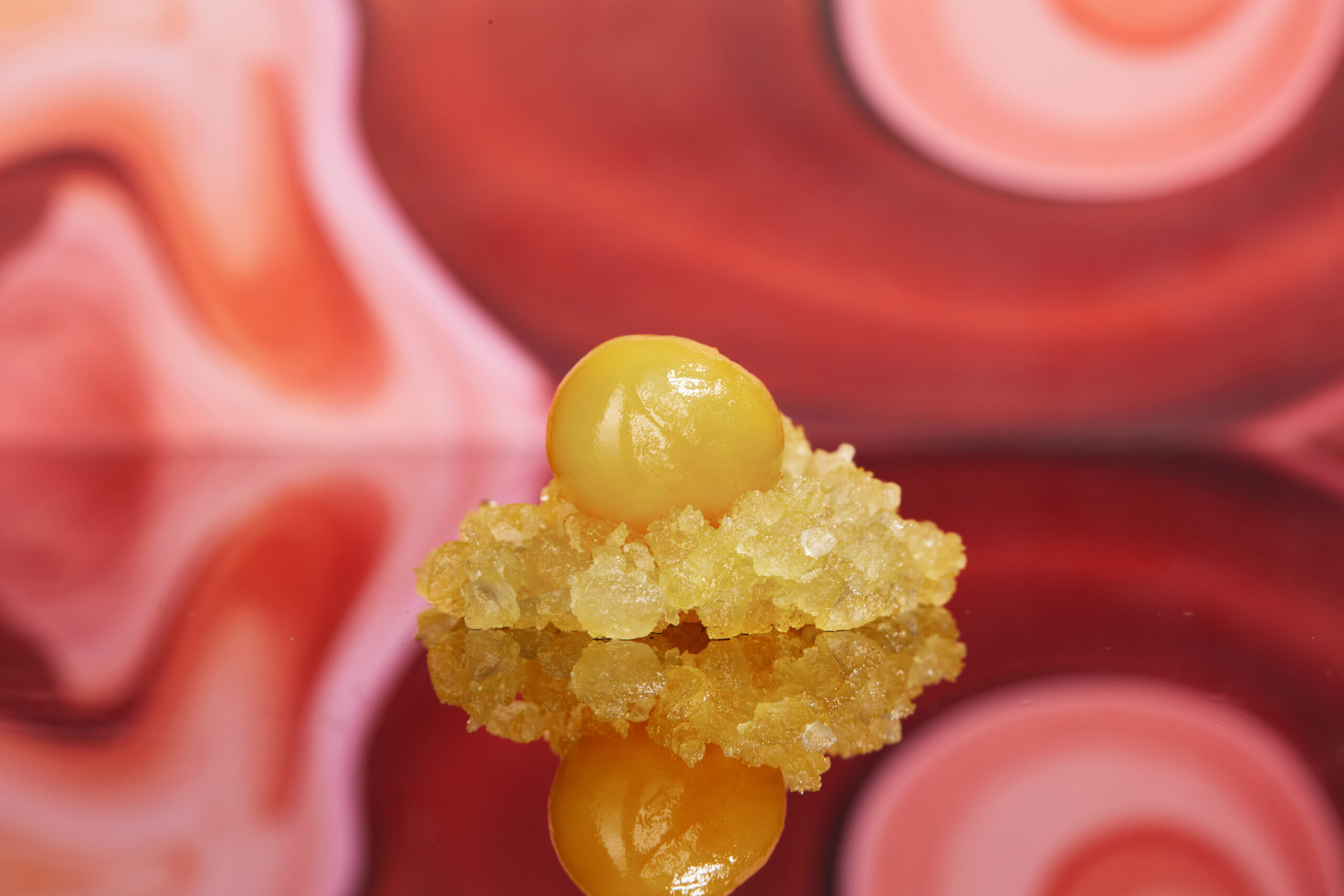
Distillate is popular due to its high potency and versatility. It can be used to dab or vaporize and can also be infused into edibles, tinctures, topicals, or added to pre-rolls, moon rocks, and various other cannabis products.
Learn more about THC distillate, how it's made, and where to find it near you.
What is THC distillate?
Distillate is a cannabis extract in which the final product has been systematically stripped of all materials and compounds except for one specific cannabinoid. The most common forms of distillate are tetrahydrocannabinol (THC) oil and cannabidiol (CBD) oil, with their percentages ranging between 90 - 100% pure cannabinoid, making them extremely potent.
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

It's the base ingredient of most edibles and vape cartridges and typically lacks flavor, taste, or aroma. And though it also lacks the beneficial terpenes that naturally occur in the source cannabis plant, it allows complete control over a final product's taste and smell. Adding terpenes to distillate later in the process is possible, and many manufacturers do this, though it's been theorized that any medicinal advantages are reduced by their initial removal.
How is distillate made?
Making distillate is a complex and technical process that requires specialized equipment, a controlled lab setting, and a deep knowledge of chemistry. Essentially, it's something you don't want to try at home. For informational purposes, the distillation process typically involves the following steps:
- Extraction: First, THC is extracted from the cannabis plant material. Chemists use various methods for the extraction process, such as carbon dioxide (CO2) extraction, ethanol extraction, or hydrocarbon extraction. Each method has its advantages and disadvantages regarding purity, efficiency, and safety.
- Winterization: This step involves mixing the raw extract with ethanol and then freezing it, which helps to separate the fats, waxes, and lipids from the cannabinoids as they coagulate and precipitate out at low temperatures.
- Filtration: The winterized solution is then filtered to remove the solidified impurities.
- Evaporation: After filtration, the solution is subjected to rotary evaporation. Heat and rotation evaporate the ethanol in this process, leaving behind a more refined cannabis oil.
- Decarboxylation: By this point, the oil is full of THCA, or tetrahydrocannabinolic acid, the non-psychoactive precursor compound to THC found in raw cannabis plants. It becomes THC when decarboxylated, typically through heat. The refined oil needs to be decarboxylated to convert THCA into psychoactive THC.
- Distillation: Finally, the decarboxylated cannabis oil is processed through a distillation unit. This process uses heat and a vacuum to vaporize and then condense the cannabinoids, separating them based on their boiling points. THC, having a unique boiling point, is isolated as a distillate.
- Refinement: Further refinement steps like molecular distillation are sometimes employed to achieve higher purity levels.
Does distillate get you high?
Whether distillate gets you high depends on the cannabinoid you're consuming. By removing virtually everything except for the desired cannabinoid, the final product is incredibly potent, so if you're consuming a THC distillate, you will probably get very high, as the resulting oil will be almost pure THC. If you consume a CBD distillate, you will tap into the therapeutic benefits of CBD, but since CBD is non-psychoactive, you will not get high.
What are the effects of THC distillate?
How you experience THC distillate depends on your individual response to THC and the dose you take.
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

Here's a list of the potential effects of THC distillate, some of which are more pleasant than others:
- Euphoria
- Relaxation
- Altered perception: Your senses, like sight and sound, might be heightened or altered.
- Increased appetite: It's common to experience the munchies, or an increase in appetite, after consuming THC.
- Dry mouth
- Red eyes
- Slower reaction times: THC may slow down your reaction times and affect motor skills, so don't drive or operate heavy machinery.
- Short-term memory loss: You might find it harder to remember things while under the influence.
- Drowsiness
- Anxiety or paranoia: In high doses, THC can lead to feelings of anxiety or paranoia.
Benefits and risks of THC distillate
As with all cannabis products, there are pros and cons to consider:
Benefits
- High potency: What can be a risk for some is a major benefit to others. THC distillate is very strong, so a little goes a long way in providing effects. It can be a cost-effective way to consume THC for those with high tolerances or medicinal needs.
- Purity: Distillate is highly refined, containing far fewer impurities and plant material than other extracts.
- Versatility: THC distillate is a flavorless oil that can be used in a variety of ways, from vaping to making edibles and topicals.
Risks
- Risk of overconsumption: THC distillate is potent by design, so it's easy to use too much, which can lead to uncomfortable effects like anxiety.
- Risk of increased tolerance: Frequently using high-THC products might lead to a higher tolerance, requiring more to achieve the same effects.
- Legal risks: It's important to know the laws regarding concentrated cannabis products in your state, as many have different limits on the amount of concentrates you can possess at a time.
- Addition of thinning agents: One of the negative points of distillate is that to make it, thinning agents have to be added to the production, including vegetable glycerine, propylene glycol, and MTC oil.
How do you use THC distillate?
There are many ways to incorporate THC distillate into your cannabis consumption routine.
Dabbing
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

Dabbing involves using a dab rig, a type of water pipe designed specifically for concentrates like distillate. You heat the nail, typically made of quartz, titanium, or ceramic, with a blow torch, drop a rice-sized amount of distillate directly onto the heated surface, and inhale the vapor produced through the mouthpiece.
Vaping
To vape THC distillate, use a vape pen designed for cannabis concentrates. Fill the chamber with a small amount (rice or pea-sized), then use the pen to heat and vaporize it. Inhale the vapor through the mouthpiece of the pen.
Making edibles
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

Since THC distillate is already activated (decarboxylated), you can directly mix it into foods or recipes. Start with a minimal amount, around 2 - 5 milligrams per dose, and ensure it's evenly distributed in the edible.
Smoking
To smoke distillate, you can spread a thin line of it over ground cannabis before rolling it into a joint. Or, mix it in with the flower in your bowl or bong for enhanced potency.
Making topicals
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

To make infused topicals, mix THC distillate with a carrier oil or lotion. Apply the mixture directly to the skin for localized effects. It's often used for therapeutic purposes like localized pain relief or skin conditions since topicals typically don't produce intoxicating effects.
Common THC distillate products
Explore some of the most common THC distillate products available in the cannabis market:
Vape cartridges and pens
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

Different brands offer distillate-filled vape cartridges and pens, which do not need loading or cleaning. They provide a hassle-free vaping experience and are ideal for discreet consumption.
Infused edibles
The category is vast. However, many brands make baked goods, gummies, chocolates, beverages, and more using only THC distillate to infuse their edibles. Any of these treats will provide a longer-lasting and often more intense effect compared to smoking or vaping, so consume them carefully and wait at least two hours after consumption to see how you're feeling before consuming more.
Infused pre-rolls
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

Many infused pre-rolls are enhanced with THC distillate. Brands will add distillate to ground cannabis before rolling it into a pre-roll, which will significantly increase its potency, making it a common choice for high-tolerance consumers.
THC distillate syringes
A THC distillate syringe is a precise, convenient way to dispense pure THC distillate. These syringes contain highly concentrated THC extract, offering an easy way to dose and apply the product. Found in most dispensaries, they're ideal for dabbing, refilling vape cartridges, or infusing edibles, and provide a mess-free, accurate way to dose THC distillate.
Tips for purchasing distillate
When purchasing THC distillate, consider the following tips to ensure you're getting the highest quality product:
- Buy from licensed dispensaries. This can ensure that a product complies with safety regulations and is free from harmful contaminants.
- Ask questions. Don't hesitate to ask your budtender questions about the product's source, extraction method, and recommended usage. Understand the type of distillate you are buying — whether it's THC, CBD, or a specific cannabinoid blend — and ensure it meets your needs and is easy to dose.
- Compare prices. While price shouldn't be the only factor, compare prices among reputable vendors to ensure you get a fair deal. Extremely low prices can sometimes indicate a low-quality product.
- Look for lab test results. Look for products that have been lab-tested and come with a certificate of analysis (COA). This provides information on potency and cannabinoid profiles and checks for pesticides, heavy metals, and solvents.
- Observe the packaging. Proper packaging is essential for maintaining the quality of the distillate. It should be well-sealed and clearly labeled with contents and usage instructions.
- Research the brand. Look for customer reviews and feedback to gauge the quality and reliability of their products.
Bottom line
Cannabis distillates are an efficient way to get high and experience specific cannabinoids in their purest form. To get the most out of distillates and be confident you're getting high-quality products, purchase THC distillate from legal brands and licensed dispensaries.
FAQ
Distillate vs. concentrates: what's the difference?
Distillate is a highly pure form of cannabis. It's usually very strong, with THC or CBD levels often exceeding 90%. Making distillate involves several steps that remove almost everything except the THC or CBD, making it flavorless. People who like distillate favor it because it's pure, potent, and flexible. You can use it in vape pens, edibles, or creams.
Concentrates cover a wider range of products like wax, shatter, and live resin. They are produced using various extraction methods, like CO2, butane, ethanol, or simple heat and pressure. Some retain more of the plant's original compounds, so concentrates can have a fuller taste and offer a variety of effects, depending on what's in them. People use concentrates primarily for dabbing or vaping, though some edible brands incorporate full-spectrum concentrates into their edibles.
What is a distillate pen?
Vape pens that contain pure distillate oil feature a cartridge filled with THC distillate, a heating element, and a battery that powers the heating element. As with any vape pen, the heating element vaporizes the oil inside the cartridge, and the vapor is then inhaled through the mouthpiece. Since they contain such a potent form of extract, these vape pens tend to be very strong.