One of the most exciting things about the cannabis plant is the way it interacts with our body's internal systems. As we learn more about its compounds, we can discover how they function with the human body.
Learn about the endocannabinoid system, how it works, and what signs may tell you if you have an endocannabinoid deficiency.
What is the endocannabinoid system?
The endocannabinoid system, or endogenous cannabinoid system (ECS), is a complex network in your body that helps maintain balance and regulate various functions like mood, sleep, appetite, and pain. Think of it as a system of signals and receivers: your body produces substances called endocannabinoids, which are like keys that fit into special locks (receptors) located throughout your body, particularly in the brain, gut, and immune system.
When these keys fit into the locks, they send messages to your cells, telling them how to respond to changes and keep everything working smoothly. This system plays a significant role in keeping your body stable and functioning properly.
How does it work?
The endocannabinoid system works through a combination of endocannabinoids, receptors, and enzymes.
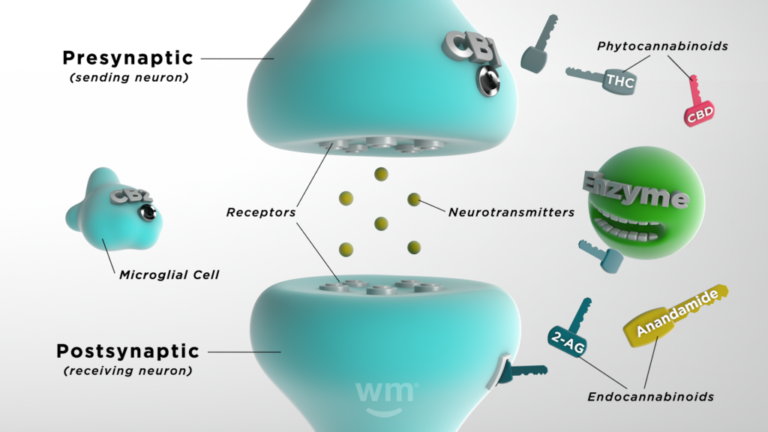 Photo by: Weedmaps
Photo by: WeedmapsImage lightbox

Here's a simplified breakdown of how it functions:
- Endocannabinoids: These molecules are similar to the cannabinoids found in cannabis plants (phytocannabinoids), but your body makes them naturally. They ensure internal functions run smoothly. Your body produces them as needed, making it tricky to know what typical levels are.
- Receptors: Found throughout your body, endocannabinoids bind to cannabinoid receptors to signal that the ECS needs to act.
- Enzymes: There are synthesizing enzymes that help turn healthy fats in your system into endocannabinoids, and there are degrading enzymes that break down the endocannabinoids once they have done their job.
Your endocannabinoid system springs into action when something brings your body out of its ideal state (homeostasis). Endocannabinoids bind to receptors, signaling that the ECS needs to take action. Depending on where the receptors are activated, the effects differ. For instance, if they're activated in a part of the brain that regulates mood, your mood might lift. If they're activated on immune cells, it might result in reduced inflammation.
What are endocannabinoids?
Endocannabinoids are small molecules that your body naturally produces. The term "endocannabinoid" can be broken down into "endo," meaning inside the body, and "cannabinoid," as they are similar to compounds found in the cannabis plant. They're part of the endocannabinoid system, which plays a crucial role in regulating various functions and processes, including mood, memory, appetite, pain, sleep quality, and immune response. Endocannabinoids work by binding to receptors (CB1 and CB2 receptors) in the endocannabinoid system that are located on almost every cell in the body, triggering various physiological processes and helping maintain homeostasis.
Two key endocannabinoids have been identified:
Anandamide (AEA)
Often referred to as the "bliss molecule," anandamide regulates pain, mood, and appetite. It's named after the Sanskrit word "ananda," which means "joy, bliss, delight."
2-arachidonoylglycerol (2-AG)
This endocannabinoid is present at higher concentrations in the brain and has roles in emotion, energy balance, immune system function, and maintaining brain health. These molecules are made from fat-like molecules within cell membranes, and they're synthesized on demand, meaning they are made exactly when needed rather than being stored and used later.
What are endocannabinoid receptors?
Endocannabinoid receptors are special proteins located on the surface of cells throughout your body. They are a critical part of the endocannabinoid system, helping to regulate various physiological processes.
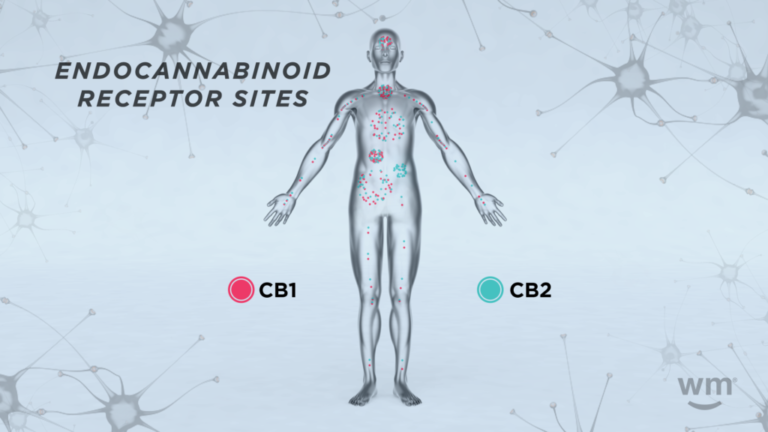 Photo by: Weedmaps
Photo by: WeedmapsImage lightbox

There are two primary types of endocannabinoid receptors:
CB1 receptors
CB1 receptors are mainly found in the brain and central nervous system (CNS) but also in some peripheral organs and tissues. They are particularly abundant in areas of the brain that control functions like mood, memory, motor control, and pain perception. When endocannabinoids or cannabinoids from external sources (like cannabis) bind to these receptors, they can influence these functions. For example, the intoxicating effects of THC are primarily due to its action on CB1 receptors in the brain.
CB2 receptors
CB2 receptors are mostly found in the peripheral nervous system, especially in immune cells. They play a significant role in managing inflammation and immune response. When endocannabinoids bind to CB2 receptors, it can signal the body to reduce inflammation, which is crucial in many diseases and conditions.
How do enzymes function in the endocannabinoid system?
In the endocannabinoid system, enzymes are like workers creating and breaking down important molecules. Some enzymes build endocannabinoids when needed, acting as tiny messengers that help keep the body balanced. Once these messengers have done their job, other enzymes break them down and clear them away, preventing too much activity from happening, which could upset the balance. It's like having a production team that makes essential components and cleans up when the work is done, ensuring everything in your body runs smoothly and stays in harmony.
When was the endocannabinoid system discovered?
The endocannabinoid system was discovered in the late 20th century through a series of research developments. The first breakthrough was in 1988 when Dr. Allyn Howlett and Dr. William Devane discovered specific receptors in the brain that respond to THC, indicating the existence of a previously unknown signaling system in the body.
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

A few years later, in 1992, researchers Dr. Raphael Mechoulam and Dr. William Devane, along with their colleague Dr. Lumír Hanuš, identified the first endocannabinoid, anandamide. This was a significant step in understanding how the body interacts with compounds like THC. Subsequent research led to the identification of other components of the endocannabinoid system, including the second major endocannabinoid, 2-arachidonoylglycerol (2-AG), and the understanding of how this system functions in regulating various physiological processes.
How does THC affect the endocannabinoid system?
THC (tetrahydrocannabinol), the intoxicating compound in cannabis, affects the endocannabinoid system by interacting with its CB1 receptors found in the brain and central nervous system and CB2 receptors throughout the body. THC also binds to some non-cannabinoid receptors.
Here's how THC influences this system:
- Mimicking endocannabinoids: On a molecular level, THC resembles endocannabinoids, specifically mimicking anandamide. Because of this similarity, THC can bind to these receptors, particularly the CB1 receptors in the brain.
- Altering brain function: When THC binds to CB1 receptors, normal brain communication alters. Brain cells (neurons) usually communicate by releasing neurotransmitters that bind to receptors on neighboring neurons. THC changes this process by binding to CB1 receptors and either increasing or decreasing the release of various neurotransmitters.
- Intoxicating effects: This interaction can lead to the intoxication commonly associated with cannabis use, such as altered perception, mood changes, impaired memory, and altered sense of time since THC affects brain areas involved in these functions.
- Tolerance: Repeated use of THC can lead to changes in the endocannabinoid system, such as reduced receptor sensitivity or density, which leads to tolerance and the need to consume more cannabis to achieve the same effect.
How does CBD affect the endocannabinoid system?
CBD (cannabidiol) affects the endocannabinoid system in a way that's quite different from THC. Unlike THC, CBD doesn't bind strongly to the cannabinoid receptors in the endocannabinoid system. Instead, its influence is more indirect and multifaceted:
- Modulating receptors: CBD interacts with various receptors in the body, either directly binding to the receptor or, in the case of cannabinoid receptors, binding indirectly, unlike the typical "lock and key" manner of THC with CB1 receptors. It can modulate the activity of the CB1 and CB2 receptors, often acting to reduce their activity. This modulation can influence how other cannabinoids, including THC, interact with these receptors.
- Enhancing endocannabinoid levels: CBD inhibits the breakdown of anandamide, an endocannabinoid, by suppressing the enzyme (fatty acid amide hydrolase or FAAH) responsible for its breakdown. This can lead to increased levels of anandamide in the body, potentially enhancing the positive effects of this endocannabinoid, like mood regulation and pain relief.
- Interaction with other systems: CBD also interacts with non-cannabinoid receptors and systems. For example, it binds to serotonin receptors, influencing mood, stress, and anxiety levels. It also affects other receptors involved in pain perception and inflammation.
- Neuroprotective and anti-inflammatory effects: CBD has demonstrated neuroprotective and anti-inflammatory properties. It can reduce inflammation in the brain and body, which is beneficial for conditions like multiple sclerosis and arthritis. Its neuroprotective properties are being researched in the context of neurodegenerative diseases.
- Reducing THC's intoxicating effects: CBD can counteract some of THC's psychoactive effects by modulating the CB1 receptor's activity. This can reduce the intensity of THC-induced anxiety or high.
What is endocannabinoid deficiency?
Endocannabinoid deficiency is a theory that suggests certain medical conditions may be caused by a deficiency or imbalance in the body's endocannabinoid system. The theory of clinical endocannabinoid deficiency (CECD) was proposed by Dr. Ethan Russo in 2004. It hypothesizes that insufficient production or functioning of endocannabinoids could lead to a range of disorders. This deficiency might be the underlying cause of some conditions that are otherwise difficult to explain or treat, such as chronic pain conditions like fibromyalgia or migraines, mood disorders like anxiety or depression, and gastrointestinal disorders like irritable bowel syndrome.
 Photo by: Gina Coleman/Weedmaps
Photo by: Gina Coleman/WeedmapsImage lightbox

The concept of endocannabinoid deficiency is still a developing area of research, but there are numerous scientific peer-reviewed publications documenting evidence of this deficiency in conditions such as autism, schizophrenia, fibromyalgia, PTSD, and others. It's not yet widely accepted as a formal diagnosis in mainstream medicine, mainly because more research is needed to fully understand the ECS and its role in these conditions. However, it's an intriguing area that could open new avenues for treating and understanding certain chronic illnesses.
Bottom line
The ECS plays a key role in maintaining internal balance and health. It consists of endocannabinoids (molecules made by your body), receptors (found throughout the body), and enzymes (which create and break down endocannabinoids). The endocannabinoid system helps regulate various functions, including mood, appetite, sleep, pain sensation, and immune responses. While substances like THC and CBD from cannabis can affect this system, the full extent of the ECS's role in health and disease is still being researched. Understanding the ECS is crucial for exploring potential treatments for various conditions and overall wellness.
FAQ
What's the difference between endocannabinoids and phytocannabinoids?
Endocannabinoids are produced naturally by the human body as part of the endocannabinoid system; they help maintain internal stability and health by interacting with cannabinoid receptors. Key endocannabinoids include anandamide and 2-AG. By contrast, phytocannabinoids are plant-derived cannabinoids found in cannabis, such as THC and CBD. Phytocannabinoids can mimic or influence the behavior of endocannabinoids, interacting with the same receptors in the body, but they come from an external source and can have different, sometimes more potent, effects.
Are there ways to stimulate your endocannabinoid system to enhance your high?
Regular exercise, foods rich in fatty acids like nuts and seeds, and some herbs and teas, such as echinacea or chamomile, may interact with the ECS and enhance the effects of a cannabis high. Yoga, meditation, and deep breathing exercises may also enhance the ECS.
Does your body produce THC?
No, your body does not produce THC. THC is a specific compound found in the cannabis plant. However, the human body does produce its own cannabinoids, known as endocannabinoids. These endocannabinoids, like anandamide and 2-arachidonoylglycerol (2-AG), share some similarities with THC in how they interact with the ECS, but they are distinct and naturally occurring compounds within the body, not derived from cannabis.
This article was reviewed by Bonni Goldstein, MD, a physician specializing in cannabis medicine in Los Angeles, California, owner and medical director of CannaCenters, and medical advisor to Weedmaps.com.